The Wurtz Fittig Reaction is basically an organic chemical reaction in which Aryl halide reacts with alkyl halides and sodium metal in the presence of dry ether which yields substituted Aromatic compounds.
The reaction is the combination of the Wurtz reaction by Adolphe Wurtz and Fittig Reaction by Rudolph Fittig. Hence the reaction is named after both the scientists.
The basic Reaction is shown below:

This reaction can also be conducted by using metals other than sodium such as potassium, iron, copper, lithium. In the case of lithium, the reaction needs to be done under ultrasound to obtain an appreciable yield.
Index
Wurtz Fittig Reaction Mechanism
The mechanism of the this reaction can be explained through 2 approaches.
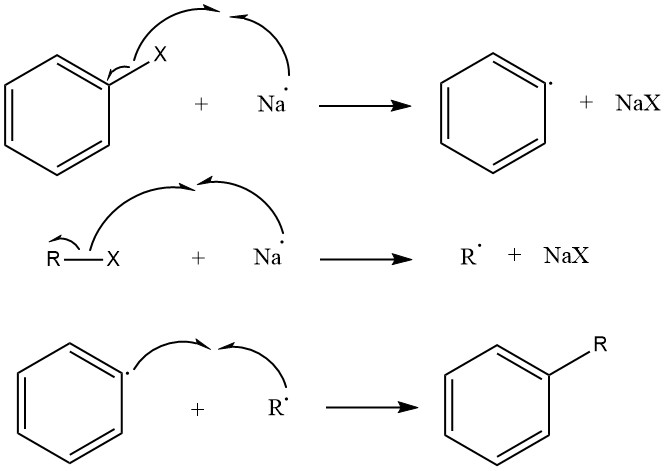
The Radical Mechanism
The first approach we will discuss is called as Radical Mechanism.
- The halogen bonds in the Aryl Halide and the Alkyl Halide are broken and hence Radicals are produced .
- The Alkyl radical and the Aryl radical react with each other which leads to the formation of a ? (Sigma) bond.
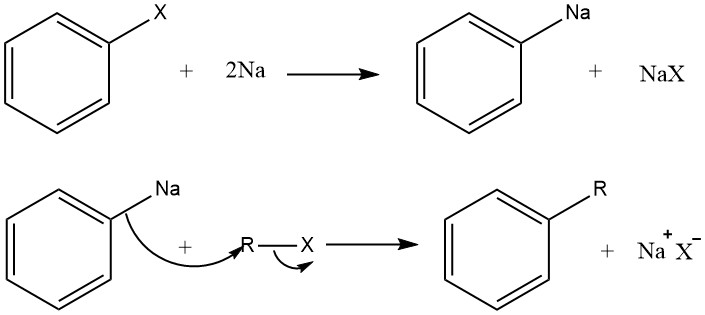
The Organo-Alkali Mechanism
And the second approach we will discuss is called as Organo-Alkali Mechanism.
In this mechanism, when the Aryl halide reacts with sodium metal an Organo-alkali intermediate compound is formed and is followed by a nucleophilic attack of the Alkyl halide. This results in the formation of Aryl-alkyl.
Applications
- This reaction is used in the laboratory synthesis of organo-silicon compounds.
- The compounds synthesized via this reaction are Calixarenes, t-ButylSilicon compounds and vinylsilanes.
- This reaction is used for the alkylation of aryl halides.
Example
Now, let us have a look at an example of this reaction mechanism.
Bromobenzene reacts with methyl bromide in presence of sodium and dry ether to form toluene.
C6H5−Br + CH3−Br +2Na ⟶ C6H5−CH3 + 2NaBr
FAQs
It is a chemical reaction in which Aryl halide reacts with alkyl halides and sodium metal in the presence of dry ether which yields substituted Aromatic compounds.
Potassium, copper, iron, lithium.
More Organic Reactions
