Zinc Phosphate (Zn3(PO4)2) is an inorganic compound that is widely used as a corrosion-resistant coating on metal surfaces. Other synonyms for the compound are tri-zinc phosphate, tri-zinc diphosphate and zinc salt.
Index
Structure & Formula
The 2D structure of the chemical compound is shown below.
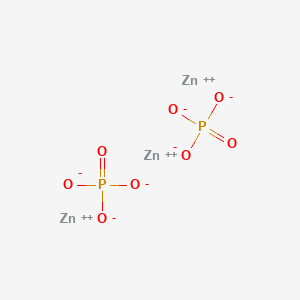
The IUPAC name for this compound is tri-zinc diphosphate and the zinc phosphate formula is given as Zn3(PO4)2.
Preparation
Zinc Phosphate is prepared by mixing magnesium oxide and zinc oxide powders. This preparation process also consists of a liquid principally of water, phosphoric acid, and buffers.
Chemical & Physical Properties
| Molecular Weight (amu) | 386.1 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 0 |
| Exact Mass of the compound | 383.69116 |
| Topological Polar Surface Area | 173 Å2 |
| Heavy Atom Count | 13 |
| Formal Charge | 0 |
| Complexity | 36.8 |
| Isotope Atom Count | 0 |
| Is it Canonicaliized? | Yes |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 5 |
| Physical Appearance | White Dry Powder |
| Density (g/cm3) | 3.998 |
| Melting Point (℃) | 900 |
| Boiling Point (℃) | 158 |
| Solubility | Insoluble in water |
Hazards
The compound is very toxic to aquatic life.
Applications
- The chemical compound is used as a corrosion inhibitor and anti-scaling agent.
- It also finds application in dyes, paint additives.
- Primer pigment also use this compound.
- The compound is also used in non-pesticidal agricultural products.
- It is one of the oldest and widely used dental cement.
- It is used for cementation of inlays, crowns, bridges, and orthodontic appliances and occasionally as a temporary restoration
FAQs
The chemical formula for the compound is Zn3(PO4)2.
The compound is primarily used for dental cement, and also for luting permanent metal and zirconium dioxide restorations.
Yes, it is a permanent cement and is considered as the standard cement to measure against.
The compound is prepared for dentistry by mixing zinc oxide and magnesium oxide powders with a liquid consisting of phosphoric acid, water, and buffers.
Related Topics:
