Zaitsev Rule is used to predict the regiochemistry of elimination reactions.
Russian chemist Alexander Zaitsev studied a number of various elimination reactions at the University of Kazan and found a general trend in the resulting alkenes.
Based on this trend, Zaitsev stated,
“The alkene formed in greatest amount is the one that corresponds to removal of the hydrogen from the alpha-carbon having the fewest hydrogen substituents.”
Index
What is Zaitsev Rule?
Zaitsev rule predicts that in an elimination reaction, the most substituted substance would be the most stable, and thus the most preferred.
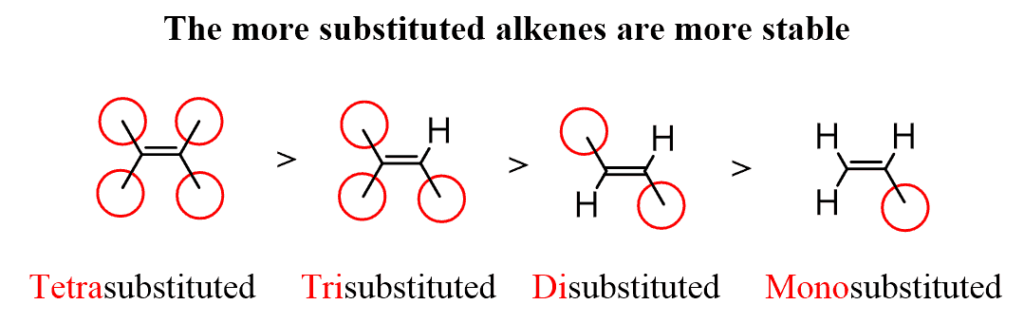
The rule doesn’t make any generalisations about the stereochemistry of the formed alkene, but only the regiochemistry of the elimination reaction.
Thermodynamic Considerations
Alkene to alkane hydrogenation is an exothermic reaction. The heat of hydrogenation, or the amount of energy released during a hydrogenation reaction, is inversely proportional to the stability of the starting alkene: the more stable the alkene, the lower the heat of hydrogenation.
Examples on Zaitsev Rule
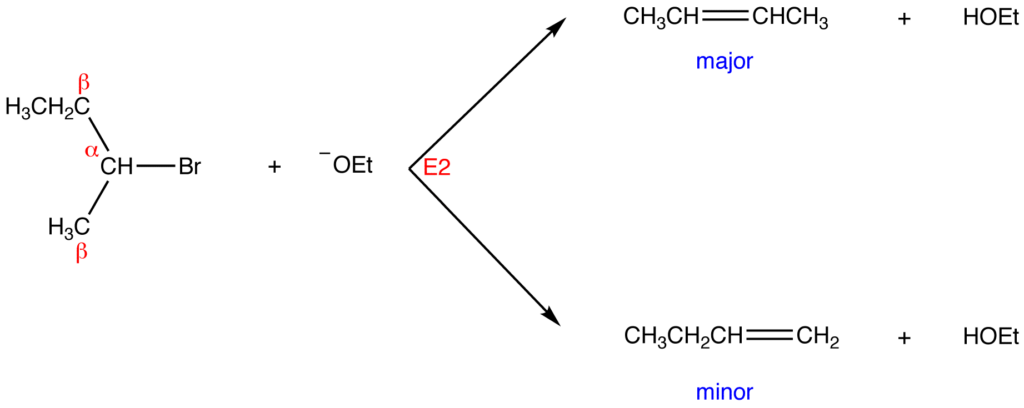
Question 1. What is the major product formed when 2-bromo-2-methylbutane is treated with sodium ethoxide in ethanol.
Answer: When 2-bromo-2-methylbutane is treated with sodium ethoxide in ethanol the major product formed is,

Question 2. Give an example to explain Zaitsev’s rule for dehydrohalogenation reaction.
Answer: When tert- Amyl chloride is treated with NaCl, H2O and HCl the major product formed is 2- Methyl-2-butene.
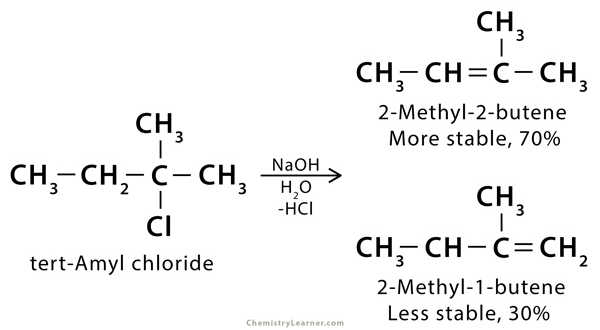
This explains Zaitsev rule.
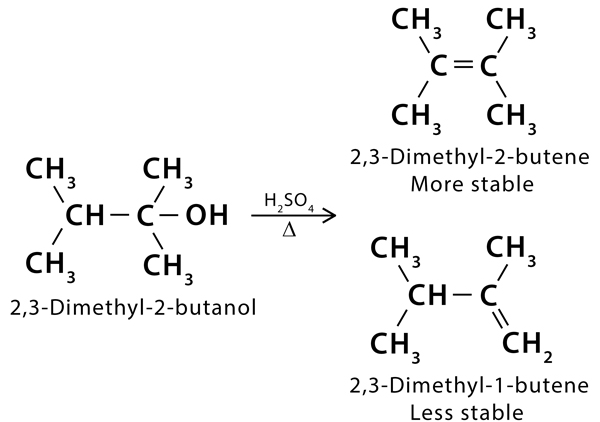
Question 3. Give an example to explain Zaitsev’s rule for dehydration reaction of aliphatic compounds.
Answer: When 2,3-dimethyl-2-butanol is treated with H2SO4 and heat the major product is 2,3-Dimethyl-2-butene .
FAQs
E1 Eliminations follow Zaitsev’s rule. With two exceptions, E2 Reactions also follow the Rule.
A Tetra Substituted Alkene is the most stable because each of the carbon forms 2 carbocation, as it is the most stable it will tend to stay as it is and is less reactive.
Carbocations are stabilized by neighboring Carbon-Carbon multiple bonds.
