Methyl Orange, also abbreviated as MeO, is an azo dye used as a pH indicator in titration. This is because, at different pH values, it gives clear and distinct colour variance.
MeO as a pH indicator gives:
Red Color – Acidic Medium, below pH 3.1.
Orange Color – Weak Acid, above 4.4.
Yellow Color – Basic Medium, basic solution.
Index
Structure, Formula & Properties
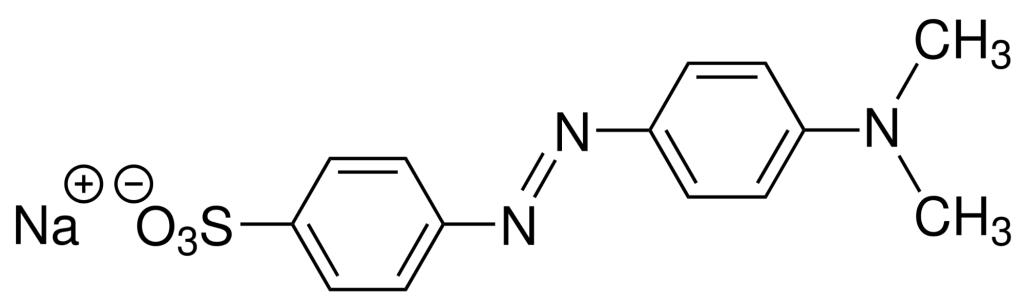
IUPAC Name – Sodium 4-{[4-(dimethylamino)phenyl]diazenyl}benzene-1-sulfonate
Molecular Formula – \(C_{14}H_{14}N_3NaO_3S\)
Molecular Weight – \(327.33 g mol^{-1}\)
Density – \(1.28 g cm^{-3}\)
Melting Point – \(300^{\circ}C\)
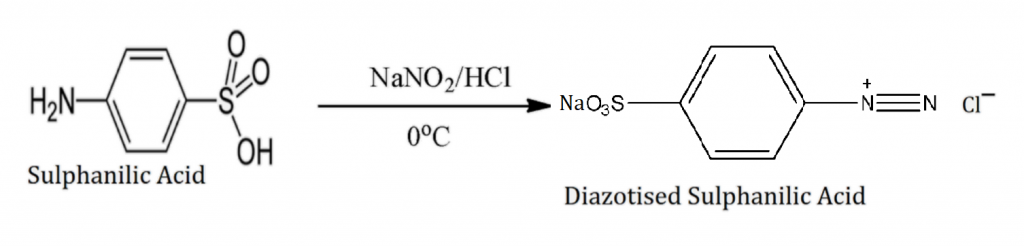
Preparation of Methyl Orange From Sulfanilic Acid
A diazonium coupling reaction is used in the production of MeO from sulfanilic acid.
Step 1: The reaction of Sulfanilic acid and Sodium Nitrate in the presence of HCl at \(0^{\circ}C\).
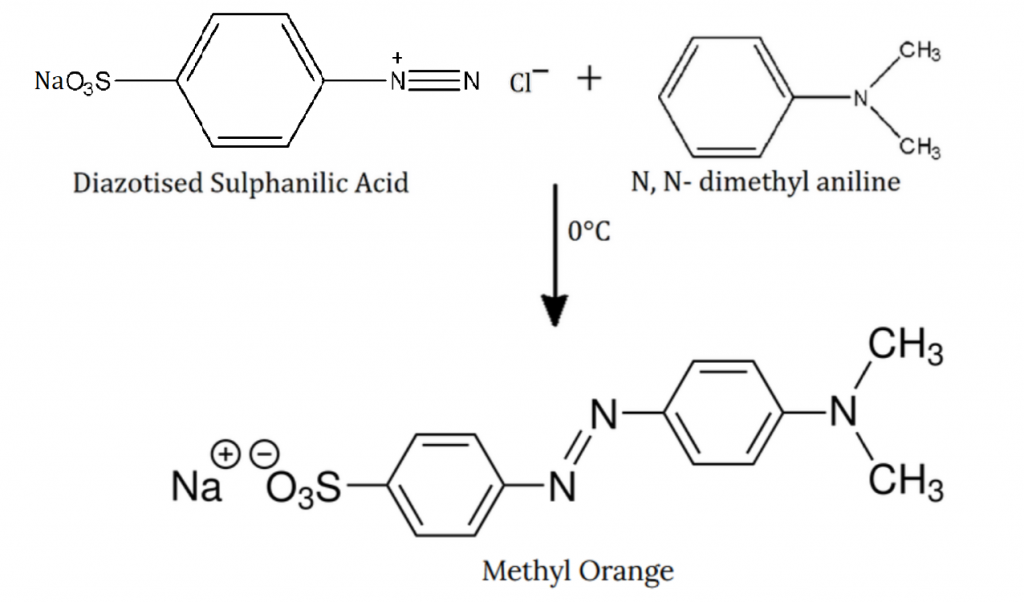
Step 2: The diazotized sulfanilic acid is treated with N,N-dimethyl aniline, at \(0^{\circ}C\).
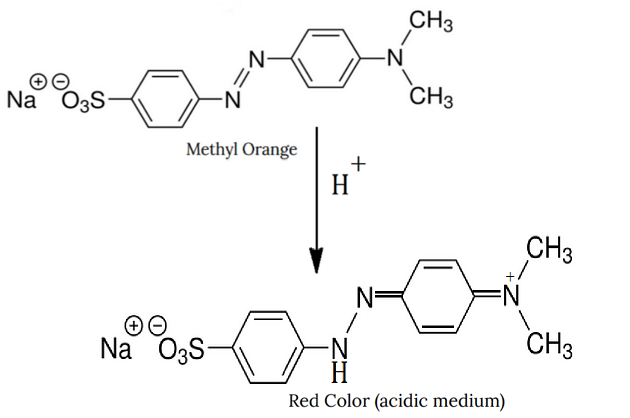
Methyl Orange in Acid
MeO gets protonated from acid at a central Nitrogen atom, which gets reduced.
It leads to a change in color from orange to Red.
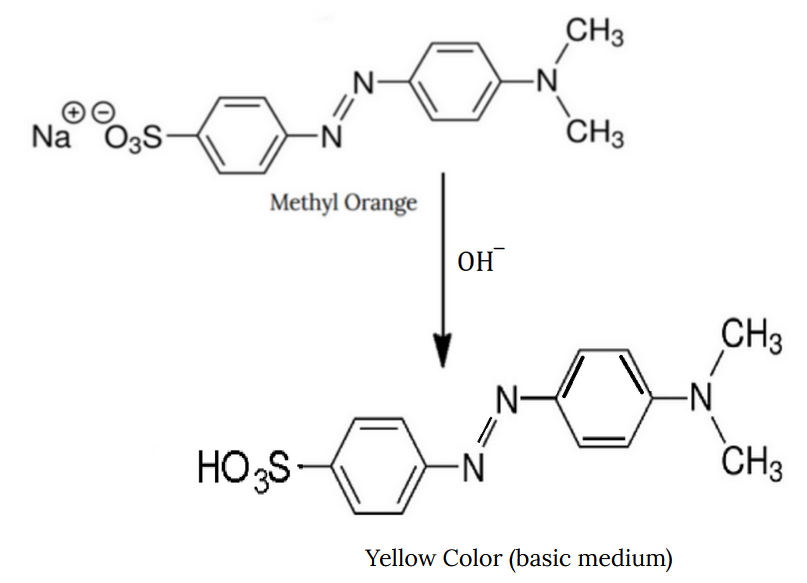
Methyl Orange in Base
MeO receives an OH– ion and forms a yellow coloration.
Applications
- They are used as an indicator for titration.
- MeO is rarely used in textiles applications because of its sensitivity to acids. It is a strongly colored compound used in dyeing and printing.
Related Topic on pH indicator:
1. Phenolphthalein
2. Thymolphthalein
Hazards
MeO has mutagenic properties i.e it can cause temporary or permanent changes in our DNA which can also lead to cancers and thus it can be carcinogenic as well in a few casses.
FAQs
MeO is an azo dye used as a pH indicator in titration.
MeO as a pH indicator gives:
Red Color – Acidic Medium, below pH 3.1.
Orange Color – Weak Acid, above 4.4.
Yellow Color – Basic Medium, basic solution.
Azo dyes are organic compounds bearing the functional group R−N=N−R′, in which R and R′ are usually aryl.
IUPAC name: Sodium 4-{[4-(dimethylamino)phenyl]diazenyl}benzene-1-sulfonate
